By Robert Cotta, Raymond Lombardi, and Robert R. Kerr, Ph.D.
The use of spherical silica gel in flash chromatography is a well-established, efficient, and rapid separation technique for the isolation and purification of organic substances whether the starting mixture comes from an organic bench chemist or a natural products extract.
Within the community of Flash Chromatography Users, there has been wide acceptance and default utilization of a standardized set of specifications used for Flash Silica. Also, there are also many chemists that have made a more conscious compromise – based on a cost-to-performance ratio to use silica for Flash Chromatography that is:
- Irregular particle sizes (commonly referred to as granular) – non-spherical
- Relatively large particles – 43-63um
- Wide particle distributions (20 microns)
- Fairly small pore sizes of 60-70 Angstroms (60-70 Å) or 6 to 7 nm
Question
Why would a user consider moving from this well-accepted standard, with its economical purchase price, to a seemingly more expensive alternative of spherical silica?
Answer
Separation efficiency.
Explanation
Depending on individual applications, efficiency can be measured through:
- Chromatographic performance
- Economical value of time to results
- Purification effectiveness
Illustration
In retrospect, there are obvious chromatographic differences when comparing standard flash grade granular/irregular shaped particles to spherical particles of smaller particle size but similar porosity. We’re going to illustrate some of these differences, right now.
These differences can provide:
- Higher loading capacities – Load more per run and finish the overall purification faster
- Faster flow rates – Total run length can be reduced
- More uniform sample distribution
- Higher plate counts
- Improved peak symmetry
- Significantly higher resolution
- Reusability – The same column can be used for successive runs
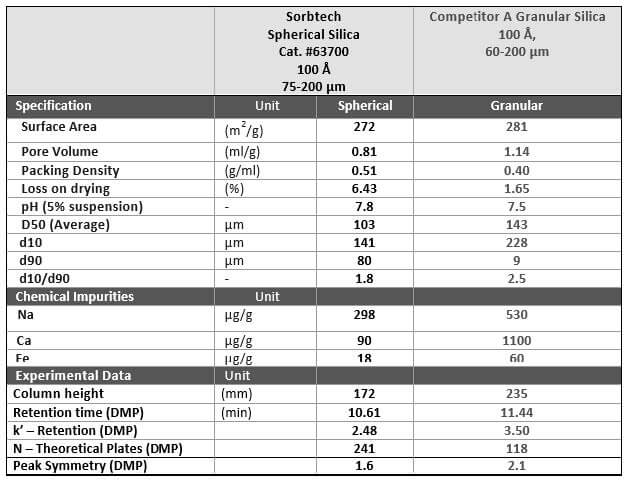
Comparison of Specifications
Table One is a comparison of our Sorbtech spherical silica gel compared to a competitor’s granular materials. As you can see, there are several chromatographic advantages that we mentioned previously in this article that can be realized by packing columns with spherical silican gel.
TABLE ONE
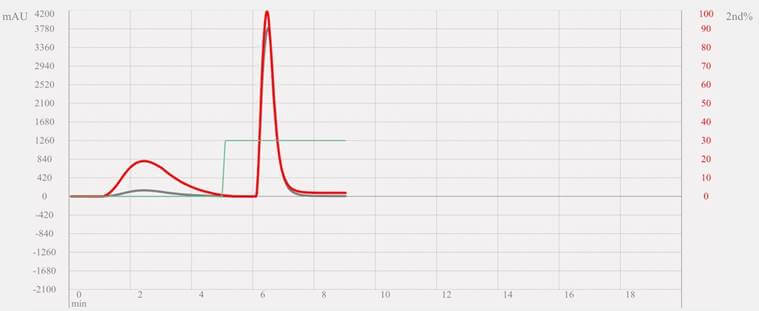
For your consideration, peak symmetry and peak resolution are also improved using spherical shaped silica due to an increase in theoretical plates (N). This next example (Figure 2) illustrates the marked advantage to more baseline resolution of the analyzed sample, as well as more symmetrical peak shape. These improvements yield purer fractions from a complex mixture that can be recovered during flash chromatography.
FIGURE TWO
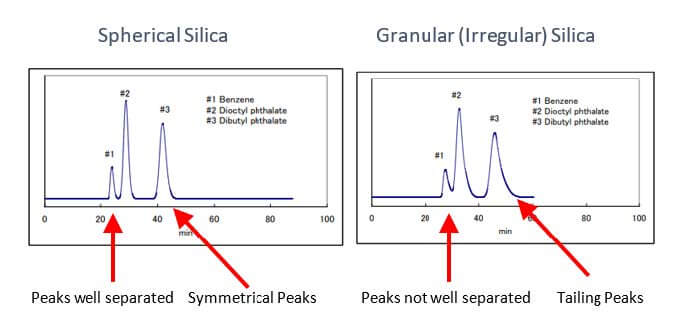
Influence of particle shape
This being said, Particle shape influences the column bed character, including the packing density and uniformity. Spherical Silica has a higher packing density than Irregular Silica.
This means:
- There is more material in the column.
• Result: you can load more material onto the column without losing resolution. - There is less liquid volume between the particles (interstitial volume).
• Result: less liquid volume for the molecules to swirl around and spread out during separation - As interstitial volume decreases
• Separation Efficiency increases
• Resolution increases
Moreover, it is MUCH easier to pack a column and obtain a homogeneous bed (no channeling) with Spherical Silica.
- Result
• lower pressure drops
• more homogenous flow profile
Another advantage of this particle shape difference is chromatographic reproducibility if all other separation conditions are held constant. The more uniform column beds formed by spherical materials provide a greater consistency from column to column and lot to lot of silica materials in packed columns. These more uniform structures can also improve the mechanical stability of the packed bed, providing a long column use life when compared to granular silica columns.
A lot more fines (very tiny particles) with granular silica
Due to the manufacturing process used to form silicas, the process for granular materials tends to generate more fines. These fines can agglomerate causing higher back pressure and accumulate as agglomerates on the column frit to contribute further to back pressure, resistance to a uniform flow
and blockage of the column outlet frit. If the particle fines are smaller in particle diameter, they can pass through the frit and contaminate the collected fractions.
It is important to keep in mind the Particle Shape is only a single parameter, the morphology parameter, of influence on the separation of compounds.
Other important factors
To optimize a separation there are several other important factors that play a role, such as:
- Solvent choice
- Temperature
- Flow rate
- Stationary Phase Chemistry
- Loading capacity
- Various other parameters not listed here
All these factors work cohesively, along with particle shape, and affect the viability of a separation.
But this takes time:
“I’m a bench chemist, not a chromatographer. I just need to get my stuff as pure as I can, as fast as I can.”
If 90-95% purity is good enough, then go for it with Irregular Silica. If you would like to routinely get 99+% purity with less effort, then use Spherical Silica.
With Spherical Silica gel:
- You can load more per run (fewer runs overall)
- Get a higher resolution
- Capture your products in a smaller volume (less to evaporate)
- Depending on your compounds, multiple uses per column
Recommendations
We highly recommend that purchase cost, traditional methodology, or legacy methods should not be the driving force behind the selection of standard, accepted flash silica specifications. Rather consider the greater separation efficiency, improved mechanical strength/lifetime, the ability for higher flow rates, and better loading capacity of higher purchase cost spherical silica to permit you to realize an improved and fiscally responsible return on your invested time and money.
More Knowledge Series Articles
HOW TO: Sample loading methods in flash chromatography
Two of the most common loading methods are a liquid sample loading and dry sample loading. Herein we briefly describe methods for both [...]
HOW TO: Step Gradients v Linear Gradients
Better, Faster Separations for Closely Eluting Peaks When compounds elute too closely together, purification becomes inefficient. That’s where gradient elution comes in. This [...]
How to properly condition a flash column
Conditioning Your Flash Column for Maximum Performance Congratulations—you’re on your way to achieving better chromatography results. When you properly condition a flash column, [...]